|
7/1/2023 0 Comments Half life chemistry Half-lives as short as 10 –6 second and as long as 10 9 years are common. Half-life is the amount of time needed for one half of a given quantity of a substance to decay. One important measure of the rate at which a radioactive substance decays is called half-life, or t 1/2. 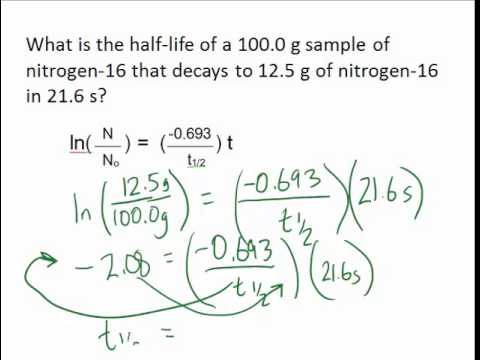
This equation shows that radioactive decay is a first-order kinetic process. The SI unit of activity is the bequerel (Bq), defined as one decay per second. R 0 is the activity (rate of decay) at t = 0. In this equation, λ is the decay constant, commonly measured in s –1 (or another appropriate unit of reciprocal time) similar to the rate law constant, k, in kinetics analyses. The mathematical description of this process is shown below.  
One type of nuclear reaction is called radioactive decay, in which an unstable isotope of an element changes spontaneously and emits radiation. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
